A) H2CO3
B) HCO3-
C) H2O
D) HPO42-
E) H2PO4-
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following yields an acidic solution when dissolved in water?
A) NO2
B) LiOH
C) K2O
D) NaCl
E) Ca(OH) 2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The pOH of a solution is 10.40.Calculate the hydrogen ion concentration in the solution.
A) 4.0 * 10-11 M
B) 3.6 M
C) 4.0 * 10-10 M
D) 2.5 * 10-4 M
E) 1.8 * 10-4 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the hydrogen ion concentration in a solution of iced tea with lemon having a pH of 2.87.
A) 2.9 * 10-2 M
B) 5.7 * 10-2 M
C) 1.3 * 10-3 M
D) 2.9 * 10-3 M
E) 5.7 * 10-4 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of ammonium chloride must be added to 250.mL of water to give a solution with pH = 4.85? [Kb(NH3) = 1.8 *10-5]
A) 4.7 g
B) 75 g
C) 2.3 * 10-3 g
D) 19 g
E) 10.g
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrosulfuric acid is a diprotic acid, for which Ka1 = 5.7 * 10-8 and Ka2 = 1 *10-19.Determine the concentration of sulfide ion in a 0.10 M hydrosulfuric solution.
A) 0.10 M
B) 7.5 * 10-5 M
C) 5.7 * 10-9 M
D) 1 * 10-19 M
E) 1 * 10-20 M
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Which of these acids is stronger, H3PO4 or H3AsO4?
Correct Answer

verified
Correct Answer
verified
Short Answer
Calculate the pH of a solution containing 0.20 g of NaOH in 2,000.mL of solution.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of 10.0 mL of 0.0020 M HCl?
A) 0.70
B) 2.70
C) 3.70
D) 5.70
E) 10.0
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of these salts will form a basic solution upon dissolving in water?
A) NaCl
B) NaNO2
C) NH4NO3
D) KBr
E) AlCl3
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.10 M NH3 solution is 1.3% ionized. Calculate the H+ ion concentration. NH3 + H2O 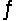 NH4+ + OH-
NH4+ + OH-
A) 1.3 * 10-3 M
B) 7.7 * 10-2 M
C) 7.7 * 10-12 M
D) 0.13 M
E) 0.10 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
The pH of a sample of river water is 6.0. A sample of effluent from a food processing plant has a pH of 4.0. What is the ratio of hydronium ion concentration in the effluent to the hydronium ion concentration in the river?
Correct Answer

verified
The hydronium ion concentratio...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Identify the conjugate base of HPO42- in the reaction HCO3- + HPO42- 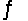 H2CO3 + PO43-
H2CO3 + PO43-
A) H2O
B) HCO3-
C) H2CO3
D) PO43-
E) None of these.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In aqueous solutions at 25°C, the sum of the ion concentrations ([H+] + [OH -])equals 1 * 10 - 14.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of an aqueous solution that contains 5.3 *1017 OH- ions per liter of solution?
A) 5.30
B) 6.06
C) 7.94
D) 8.70
E) 11.3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a solution prepared by mixing 10.0 mL of a strong acid solution with pH = 2.00 and 10.0 mL of a strong acid solution with pH = 6.00?
A) 2.0
B) 2.3
C) 4.0
D) 6.0
E) 8.0
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the formula for the conjugate base of H2PO4-.
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the chemical formula for nitric acid.
Correct Answer

verified
Correct Answer
verified
Short Answer
Lime is used in farming to reduce the acidity of the soil. The chemical name for lime is calcium oxide. When water in the soil reacts with lime, what base is formed?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
P4O10 is classified as an acidic oxide because it
A) reacts with acids to produce a salt.
B) is insoluble in water.
C) reacts with water to produce OH-.
D) gives a solution of phosphoric acid, H3PO4, on dissolving in water.
E) can act as a Lewis base by donating electron pairs.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 163
Related Exams