A) Sr
B) Rb
C) Na
D) Ca
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name for the group of elements indicated by the shaded portion of the periodic table? 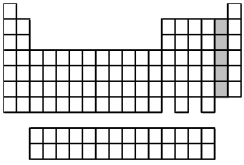
A) alkaline earth metals
B) group 3A elements
C) halogens
D) noble gases
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
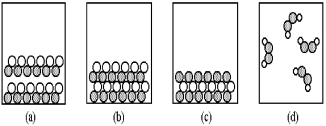 -Which of the above pictures are more likely to represent ionic compounds?
-Which of the above pictures are more likely to represent ionic compounds?
A) pictures (a) and (b)
B) pictures (a) and (d)
C) pictures (b) and (c)
D) pictures (b) and (d)
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium,strontium,and barium are all prepared commercially by the same method which is
A) electrolysis of the molten metal oxides.
B) electrolysis of the molten metal halides.
C) chemical reduction of the metal oxides with aluminium.
D) chemical reduction of the metal halides with oxygen.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which alkaline earth metal reacts the most vigorously with water at room temperature?
A) Be
B) Ca
C) Ba
D) Sr
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The four spheres below represent K+,Ca2+,Cl-,and S2-,not necessarily in that order. 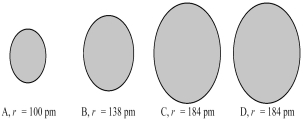 -Which sphere most likely represents the S2- ion?
-Which sphere most likely represents the S2- ion?
A) A
B) B
C) A or B
D) C or D
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following reactions is inconsistent with the chemistry of the halogens?
A) H2(g) + Cl2(g) → 2 HCl(g)
B) Fe(s) + F2(g) → FeF2(s)
C) Br2(l) + 2 Cl-(aq) → 2 Br-(aq) + Cl2(g)
D) I2(s) + 3 Cl2(g) → 2 ICl3(s)
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ionic compound would be expected to have the highest lattice energy?
A) NaCl
B) MgO
C) AlF3
D) Al2O3
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which species does not have an octet of electrons for its outer core?
A) C4-
B) P3-
C) O2-
D) Mg+
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Of the following,which element has the highest first ionization energy?
A) Cl
B) F
C) O
D) S
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which ionization process requires the most energy?
A) O(g) → O+(g) + e-
B) O+(g) → O2+(g) + e-
C) F(g) → F+(g) + e-
D) F+(g) → F2+(g) + e-
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is not generally considered to be a chemical reaction of the alkaline earth metal calcium?
A) Ca(s) + Cl2(g) → CaCl2(s)
B) 2 Ca(s) + 2 H2O(l) → 2 Ca(OH) 2(aq) + H2(g)
C) 6 Ca(s) + 2 N2(g) → 2 Ca3N2(s)
D) Ca(s) + O2(g) ��→ CaO2(s)
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following pictures represent alkali halide salts. 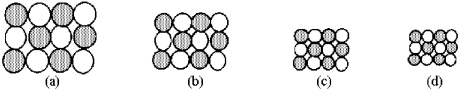 -Which salt has the lowest lattice energy?
-Which salt has the lowest lattice energy?
A) picture (a)
B) picture (b)
C) picture (c)
D) picture (d)
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ionic compounds would be expected to have the highest lattice energy?
A) LiCl
B) NaCl
C) KCl
D) RbCl
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The four spheres below represent Na+,Mg2+,F⁻,and O2-,not necessarily in that order. 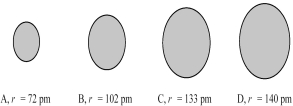 -Which sphere most likely represents the F- ion?
-Which sphere most likely represents the F- ion?
A) A
B) B
C) C
D) D
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An element that has the valence electron configuration 3s23p3 belongs to which period and group?
A) period 3;group 3A
B) period 3;group 5A
C) period 4;group 3A
D) period 4;group 5A
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the product(s) when the reactants Be(s) + Br2(l) are mixed.
A) BeBr(s)
B) BeBr2(s)
C) Be2Br(s)
D) BeBr3(s)
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
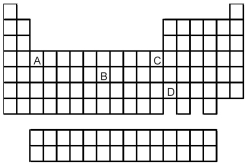 -Which element,indicated by letter on the periodic table above,has a 3+ ion with the electron configuration 1s2 2s2 2p6 3s2 3p6?
-Which element,indicated by letter on the periodic table above,has a 3+ ion with the electron configuration 1s2 2s2 2p6 3s2 3p6?
A) A
B) B
C) C
D) D
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
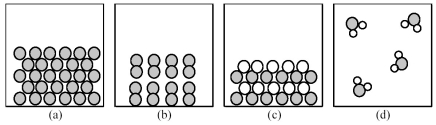 -Which of the above pictures best represents a gaseous covalent compound?
-Which of the above pictures best represents a gaseous covalent compound?
A) picture (a)
B) picture (b)
C) picture (c)
D) picture (d)
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which contains covalent bonds?
A) NaH and HCl
B) only HCl
C) only NaCl
D) only NaH
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 173
Related Exams